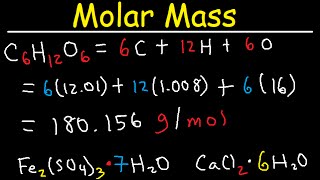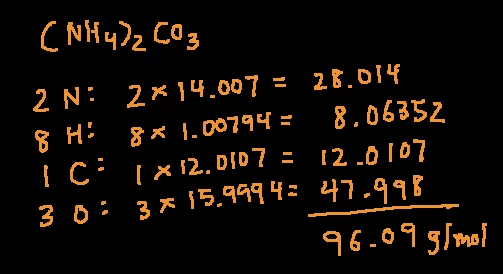Work Problem: Calculation of Molar Mass of Unknown Base Using Titration With a Strong Acid | Hey Chemistry
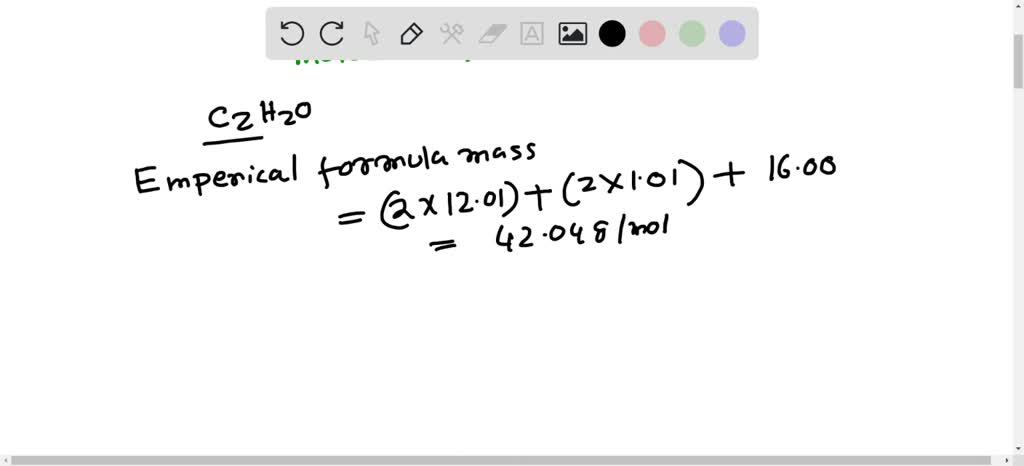
SOLVED: Calculate the molecular formula of a compound with the empirical formula C2H2O and a molar mass of 126.12 g/mol.

Question Video: Identifying the Formula of a Compound When Given a Balanced Chemical Equation and the Formula Mass | Nagwa