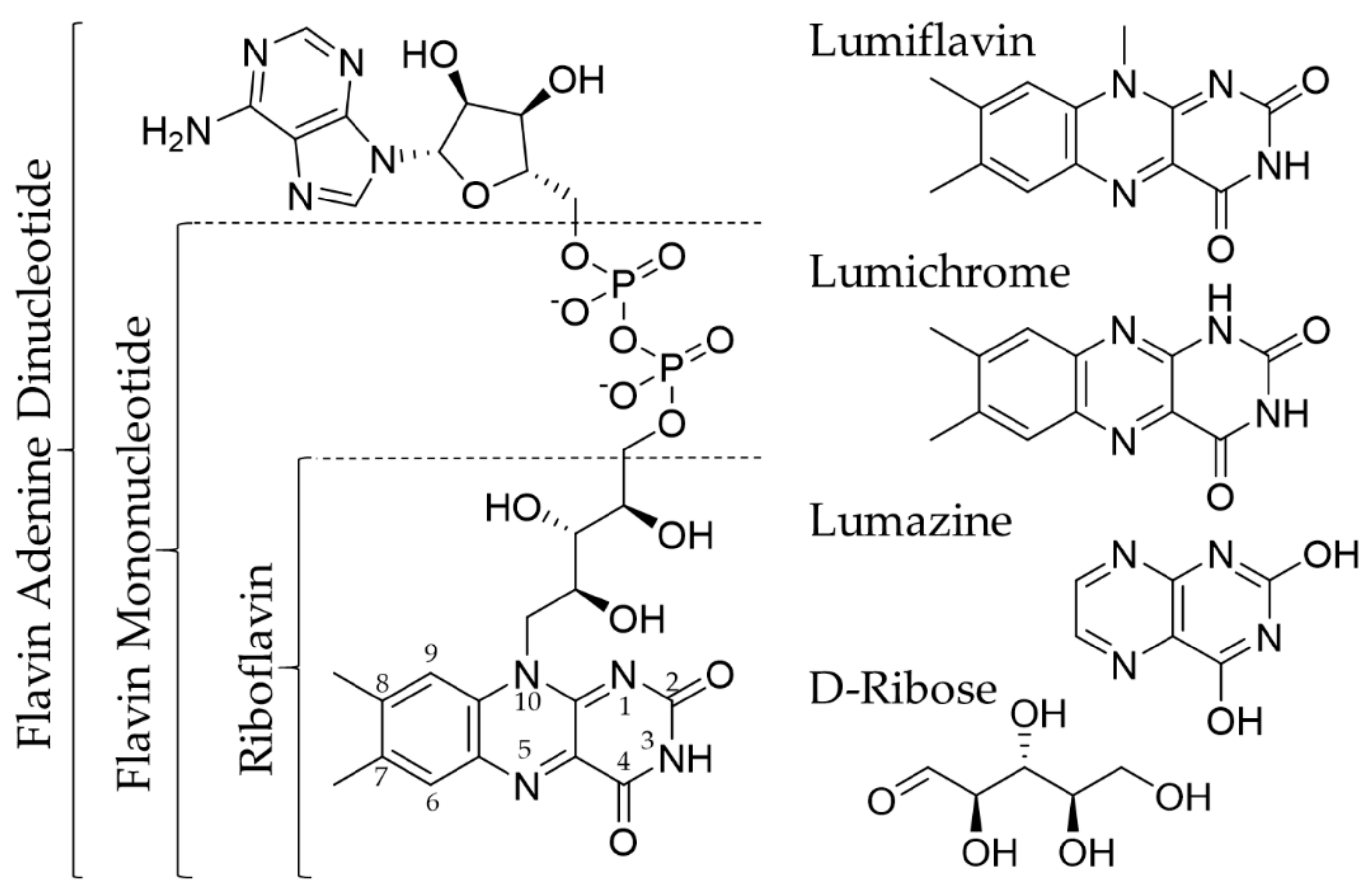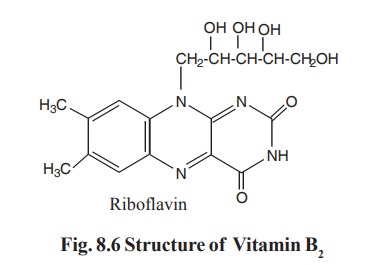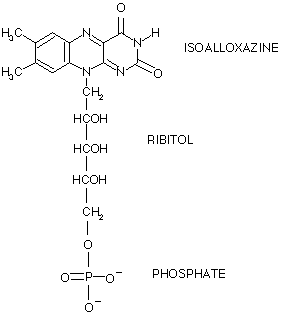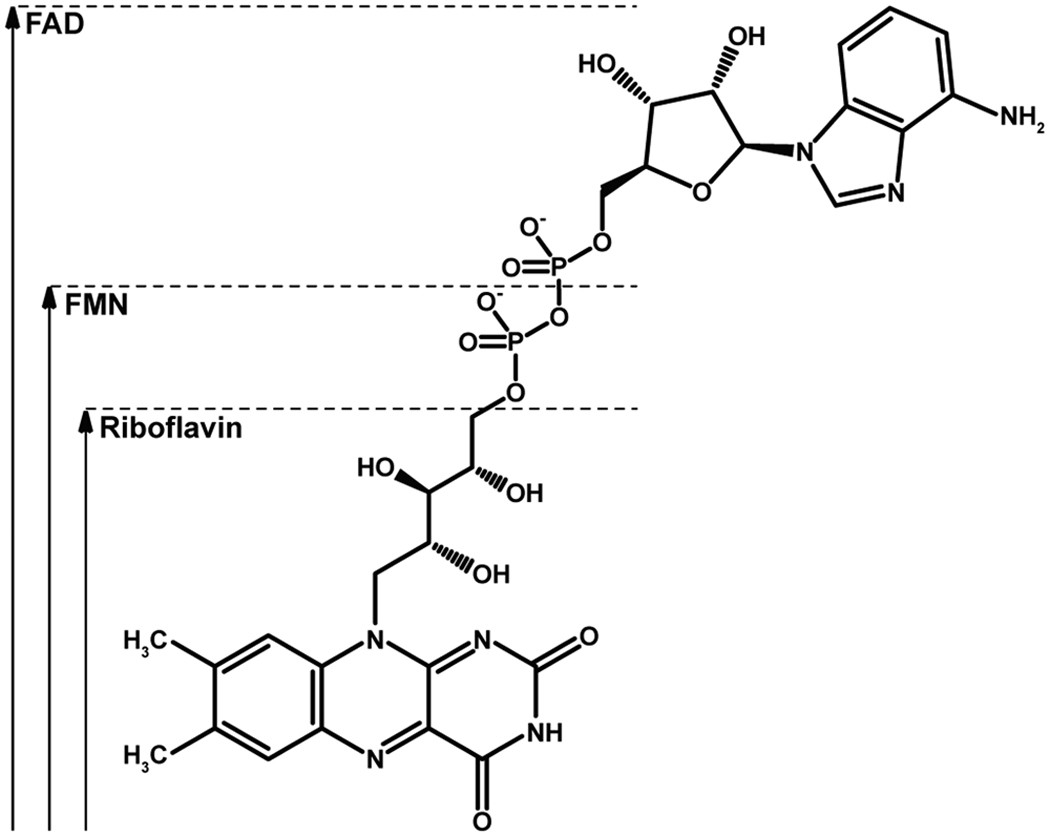
Proline dehydrogenase from Thermus thermophilus does not discriminate between FAD and FMN as cofactor | Scientific Reports

Figure 1 from Active site probes of flavoproteins. Determination of the solvent accessibility of the flavin position 8 for a series of flavoproteins. | Semantic Scholar
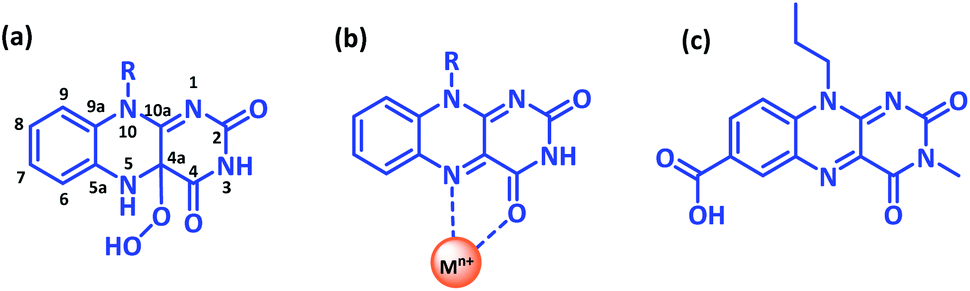
Modulating catalytic activity of a modified flavin analogue via judicially positioned metal ion toward aerobic sulphoxidation - RSC Advances (RSC Publishing) DOI:10.1039/D1RA06558K

SOLVED: The reactive part of FAD is and the reactive part of NAD+ is an isoalloxazine ring; a nicotinamide ring b) niacin; an isoalloxazine ring an isoalloxazine ring; niacin nicotinamide ring; an

SOLVED: (Q47) Which of the following statements is correct regarding what happens during the reduction of FAD? A flavin group is transferred An equivalent of hydride ion is transferred The isoalloxazine ring
The ins and outs of vanillyl alcohol oxidase: Identification of ligand migration paths | PLOS Computational Biology

Isoalloxazine Ring of FAD Is Required for the Formation of the Core in the Hsp60-assisted Folding of Medium Chain Acyl-CoA Dehydrogenase Subunit into the Assembly Competent Conformation in Mitochondria (∗) - Journal
3 Structure of isoalloxazine ring system with ribityl chain where X =-H... | Download Scientific Diagram
Electronic transitions in the isoalloxazine ring and orientation of flavins in model membranes studied by polarized light spectroscopy | Biochemistry

Solid-State Structural Properties of Alloxazine Determined from Powder XRD Data in Conjunction with DFT-D Calculations and Solid-State NMR Spectroscopy: Unraveling the Tautomeric Identity and Pathways for Tautomeric Interconversion | Crystal Growth &

PDF) Theoretical analysis of the electron spin density distribution of the flavin semiquinone isoalloxazine ring within model protein environments | Jesus Martinez - Academia.edu

Flavogenomics – a genomic and structural view of flavin‐dependent proteins - Macheroux - 2011 - The FEBS Journal - Wiley Online Library