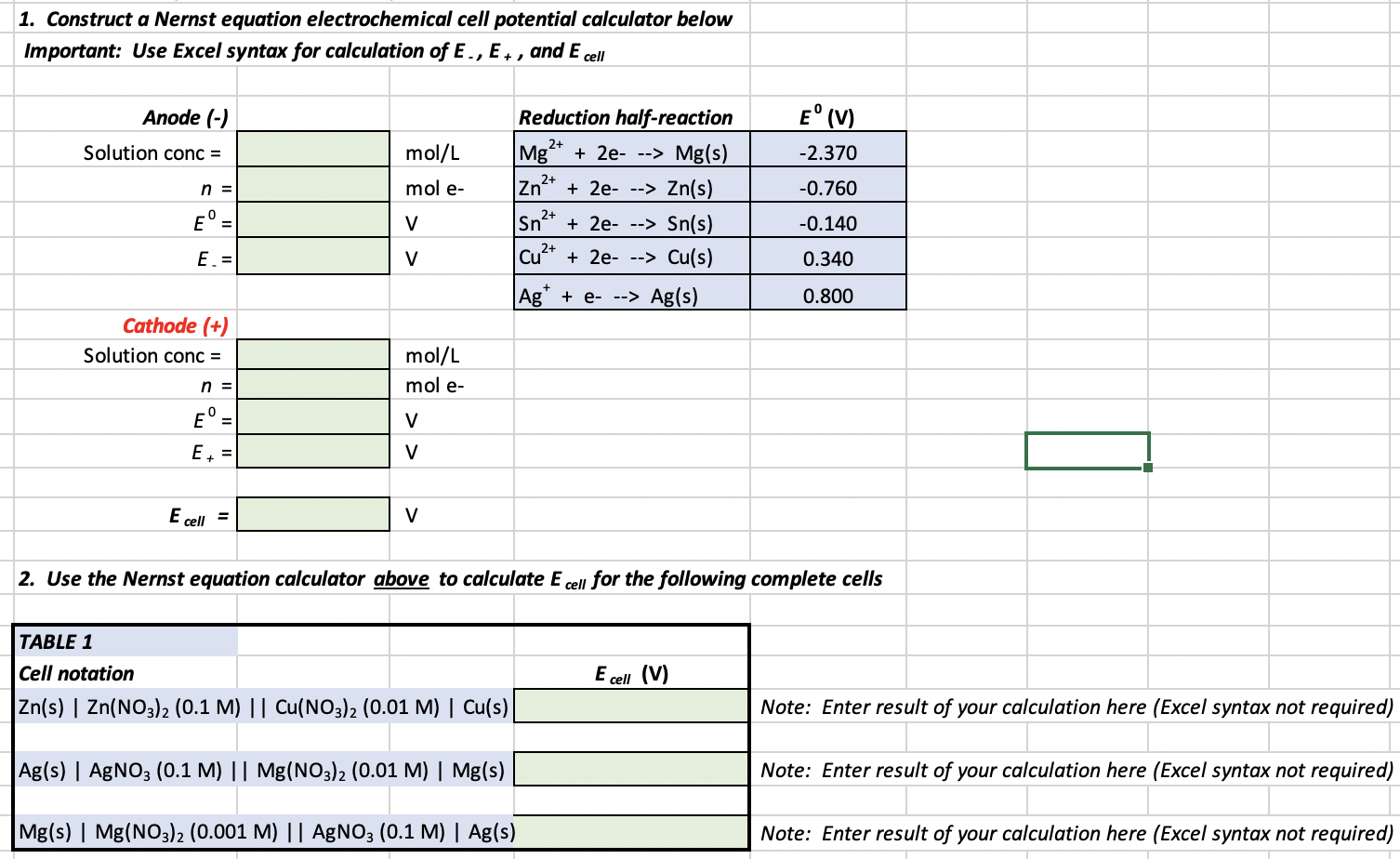
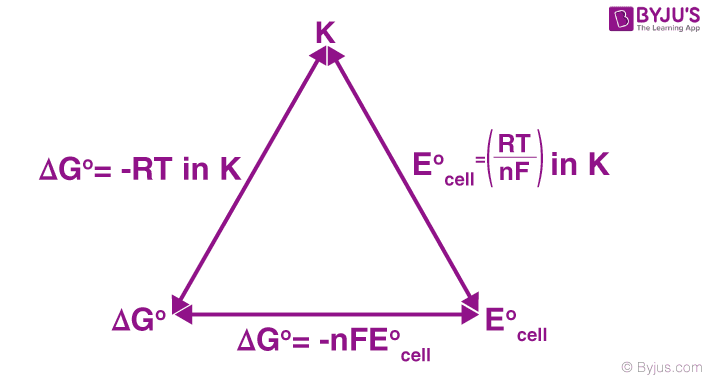
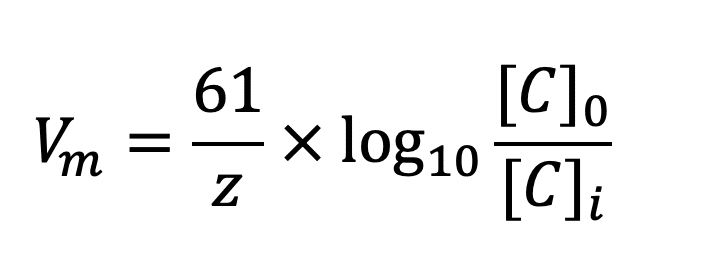SOLVED: Part C: Concentration Determination of an Unknown Using the Nernst Equation 1. One cell will serve as the reference electrode which is 0.10 M Zn2+/Zn, as in part A Obtain samples

Numerical calculations: Nernst equilibrium potential calculator. The... | Download Scientific Diagram



-437.png)